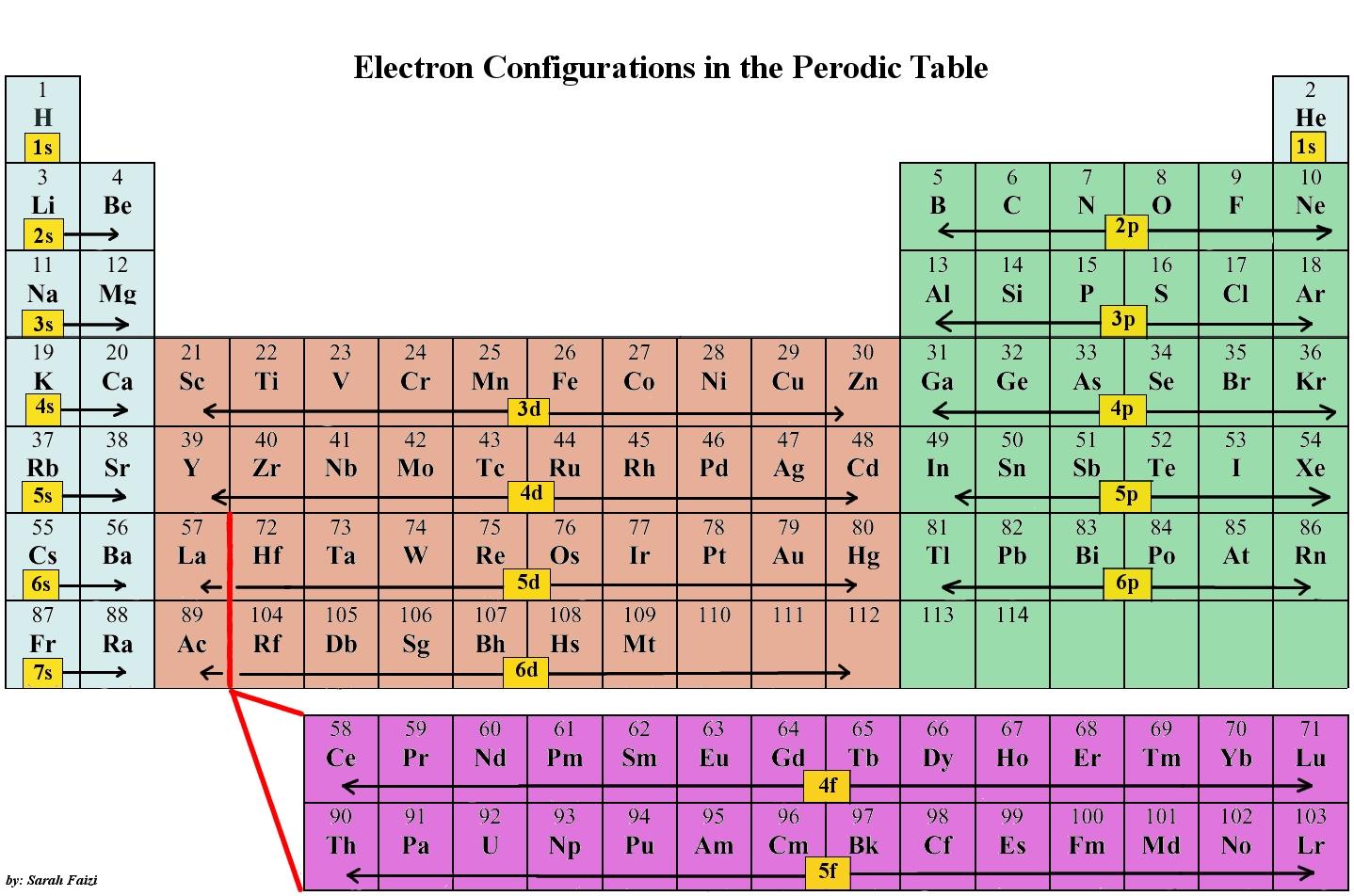
Printable Periodic Table Electronegativity Configuration: A Comprehensive Guide
What is Electronegativity?
The periodic table is a fundamental tool in chemistry, and understanding its various configurations is crucial for students and professionals alike. One of the most important configurations is the electronegativity configuration, which shows the ability of an atom to attract electrons in a covalent bond. A printable periodic table electronegativity configuration can be a valuable resource for those looking to learn more about chemical properties and reactions.
The electronegativity of an atom is a measure of its ability to attract electrons, and it is typically measured on the Pauling scale. The higher the electronegativity value, the more likely an atom is to attract electrons. By understanding the electronegativity configuration of the periodic table, chemists can better predict the behavior of atoms in different compounds and reactions.
How to Use the Printable Periodic Table
What is Electronegativity? Electronegativity is a measure of an atom's ability to attract electrons in a covalent bond. It is an important concept in chemistry, as it helps predict the behavior of atoms in different compounds and reactions. The electronegativity configuration of the periodic table shows the relative electronegativity values of different atoms, allowing chemists to compare and contrast their properties.
How to Use the Printable Periodic Table To get the most out of a printable periodic table electronegativity configuration, it's essential to understand how to use it. Start by downloading and printing the table, then use it to identify the electronegativity values of different atoms. You can use this information to predict the behavior of atoms in different compounds and reactions, and to better understand the chemical properties of various elements. With a printable periodic table electronegativity configuration, you'll have a valuable resource at your fingertips to help you learn and understand chemistry.